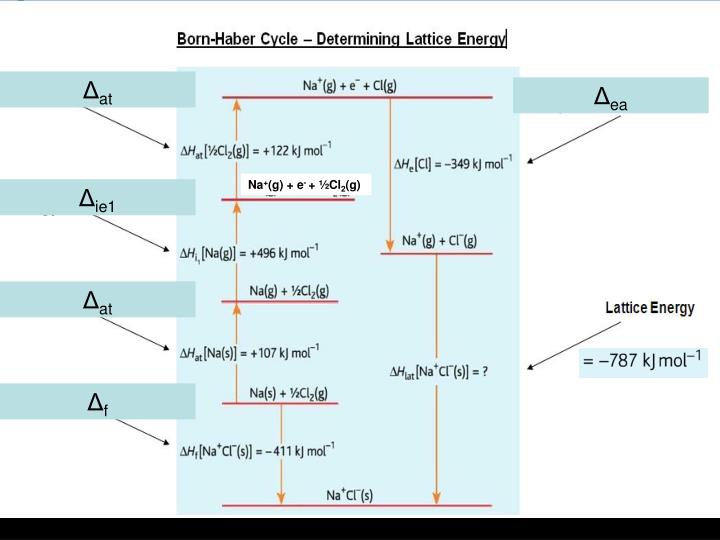 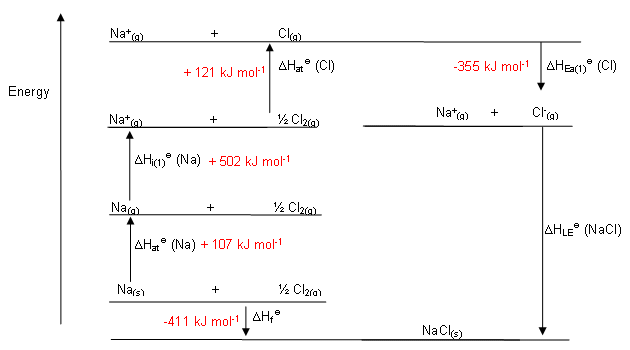
The magnitude of the forces that hold an ionic substance together has a dramatic effect on many of its properties. The Relationship between Lattice Energies and Physical Properties The order of increasing lattice energy is RbCl ZnS > SrSe > LiCl > KBr Because the cation and the anion in BaS are both larger than the corresponding ions in CaO, the internuclear distance is greater in BaS and its lattice energy will be lower than that of CaO. Because Ba 2 + lies below Ca 2 + in the periodic table, Ba 2 + is larger than Ca 2 +. To decide whether BaS or CaO has the greater lattice energy, we need to consider the relative sizes of the ions because both compounds contain a +2 metal ion and a −2 chalcogenide ion. Consequently, we expect RbCl, with a (−1)(+1) term in the numerator, to have the lowest lattice energy, and GaP, with a (+3)(−3) term, the highest. We know from Equation 4.4 that lattice energy is directly proportional to the product of the ionic charges. The compound GaP, which is used in semiconductor electronics, contains Ga 3 + and P 3− ions the compound BaS contains Ba 2 + and S 2− ions the compound CaO contains Ca 2 + and O 2− ions and the compound RbCl has Rb + and Cl − ions. For compounds with ions with the same charge, use the relative sizes of the ions to make this prediction. Using Equation 4.2.1, predict the order of the lattice energies based on the charges on the ions. 
A similar effect is seen when the anion becomes larger in a series of compounds with the same cation.Īrrange GaP, BaS, CaO, and RbCl in order of increasing lattice energy.Īsked for: order of increasing lattice energy Because r 0 in Equation 4.2.1 is the sum of the ionic radii of the cation and the anion ( r 0 = r + + r −), r 0 increases as the cation becomes larger in the series, so the magnitude of U decreases. This effect is illustrated in Figure 4.2.2, which shows that lattice energy decreases for the series LiX, NaX, and KX as the radius of X − increases. For example, the calculated value of U for NaF is 910 kJ/mol, whereas U for MgO (containing Mg 2 + and O 2− ions) is 3795 kJ/mol.īecause lattice energy is inversely related to the internuclear distance, it is also inversely proportional to the size of the ions. Source: Data from CRC Handbook of Chemistry and Physics (2004).īecause the lattice energy depends on the product of the charges of the ions, a salt having a metal cation with a +2 charge (M 2 +) and a nonmetal anion with a −2 charge (X 2−) will have a lattice energy four times greater than one with M + and X −, assuming the ions are of comparable size (and have similar internuclear distances). Note that r 0 may differ between the gas-phase dimer and the lattice. In such an arrangement each cation in the lattice is surrounded by more than one anion (typically four, six, or eight) and vice versa, so it is more stable than a system consisting of separate pairs of ions, in which there is only one cation–anion interaction in each pair. While Equation 4.1.1 has demonstrated that the formation of ion pairs from isolated ions releases large amounts of energy, even more energy is released when these ion pairs condense to form an ordered three-dimensional array. These properties result from the regular arrangement of the ions in the crystalline lattice and from the strong electrostatic attractive forces between ions with opposite charges. They are not easily deformed, and they melt at relatively high temperatures. 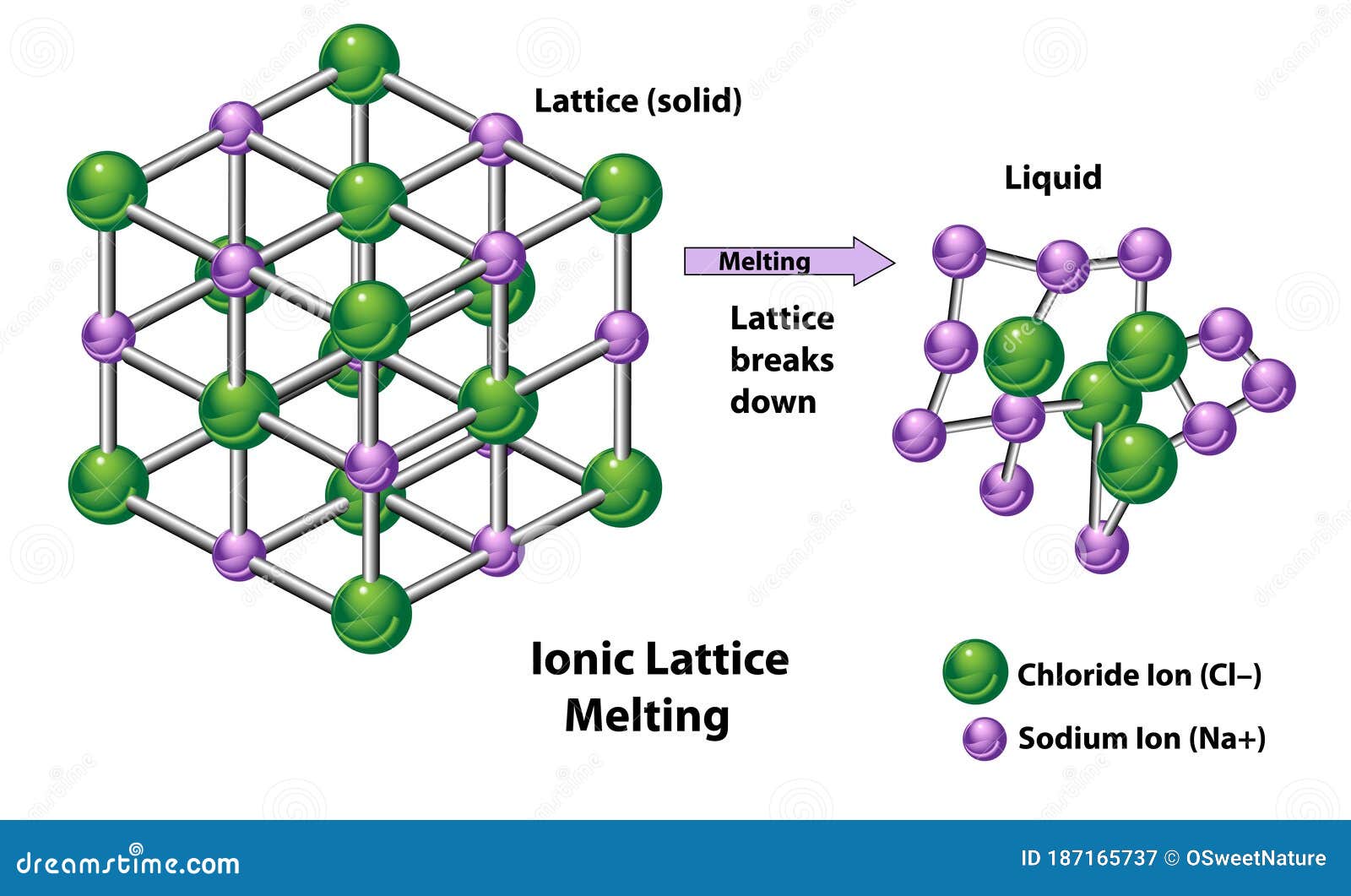
Ionic compounds are usually rigid, brittle, crystalline substances with flat surfaces that intersect at characteristic angles. Metal ores are commonly combinations of metal atoms with oxygen atoms, and this combination is produced when metals rust, so the process where electrons are transferred to the oxygen atoms from the metal atoms is known as oxidation of the metal and the reverse process, where pure metals are produced is called reduction of the ore to the metal. The reaction of a metal with a nonmetal usually produces an ionic compound that is, electrons are transferred from the metal to the nonmetal. To understand the relationship between the lattice energy and physical properties of an ionic compound.
0 Comments
If you only want the print driver (without the Photosmart Software Suite), it is available as a. Our Community is comprised of volunteers - people who own and use HPdevices. This download includes the HP Photosmart Software Suite and Driver. Thank you for participating in the HP Community. NOTE: Content depends on device type and Operating SystemĬategories: Alerts, Access to the Print and Scan Doctor (Windows), Warranty Check, HP Drivers / Software / Firmware Updates, How-to Videos, Bulletins/Notices, Lots of How-to Documents, Troubleshooting, Manuals > User Guides, Product Information (Specifications), Supplies, and more When the website support page opens, Select (as available) a Category > Topic > Subtopic HP Printer Home Page - References and Resources – Learn about your Printer - Solve Problems Printer Specifications for HP Photosmart e-All-in-One (D110a and D110b) Printers Will this work? No idea - I don't have a way to test the setup. Printer Wi-Fi Direct > Ad-Hoc > Wireless without the use of a router HP printers - Windows built-in driver fails to install If it is not listed, go to Windows Scan (in English) to install it. Windows Scan app: In Windows, search for and open the Windows Scan app.Windows Paint: In Windows, search for and open Paint.Click New Scan, and then select your printer. Windows Fax and Scan: In Windows, search for and open Windows Fax and Scan. 
Scan documents and photos: Complete scan jobs with a Windows app that works with the built-in driver. Scan? See Section FAQ > How do I use the printer functions with the built-in driver? HP Printers - Installing and Using the Windows Built-in Print Driver Windows 10 > Option to install the Windows Built-in Print Drivers.

Superior support includes knowledgeable regional managers that ensure installers have been thru a company training program that includes estimating.There are several crucial areas of support an ICF manufacturer should provide. When selecting the best ICF building system for a project, it is vital to choose a manufacturer and dealer that offers superior support. The Best ICF Block Systems Provide Excellent Manufacturer Support The planks are connected with brackets or ties.įive Tips for Choosing the Best ICF Block Systemsġ. Plank systems are 8-inch to 12-inch tall and 4-feet to 8-feet long.They are connected with brackets or ties. Panel system units are 1-foot to 4-foot tall and 4-foot to 8-foot long, with a flat rectangular shape, like a wall.Blocks typically are 12-inch to 16-inch tall and 4-feet long. Block systems are hollow-core blocks that stack and interlock like Legos.The most common type of ICFs are hollow blocks however, there are also panel and plank ICF systems. CI significantly lessens thermal bridging through the wall. Steel rebar or steel fibers strengthen the reinforced concrete filled hollow centers, which creates a layer of continuous insulation (CI). ICFs are stacked to form the shape of the walls. What are Insulated Concrete Form (ICF)Įxpanded polystyrene insulation (EPS) make-up insulated concrete forms (ICFs). Fox Block ICF excels in all these areas, which makes it an excellent choice for the best ICF block system. Important considerations when choosing the best ICF block product include excellent manufacturer and dealer support, regional manufacturing and local inventory, code compliance, easy installation, and energy-efficiency. Chief among the changes is shared Tab Groups. MacOS Ventura will introduce a number of new tricks to Apple’s standard suite of programs, including Safari. Social browsing with shared Tab Groups in Safari You’ll also be able to group windows together in the sidebar – handy for swapping between multi-app tasks in a jiffy. One click will be all it takes to switch. The idea is to provide an instant overview of all the windows you’re working on, rather than layering them behind each other. Your main window will still be displayed front and centre, but other open app windows will now appear as thumbnails on the left of your display. Stage Manager – new for macOS Ventura – aims to streamline the art of multi-tasking. Gestures might simplify app switching, but it remains all too easy to drown in windows. The most recent MacBook Air and 13in MacBook Pro models, which are the first with Apple’s new M2 silicon, launched with Monterey, but will be first in line for Ventura when it gets officially released. Wondering if your Mac makes the cut? Check the list below to see which devices will support Ventura. All Cupertino computers come with a shelf-life for updates – and MacOS Ventura doesn’t do anything to change that pattern. While your older Mac might still be going strong, that doesn’t mean it will support Apple’s latest software release. It does not store any personal data.Ventura is available as a free download for compatible Apple laptops and desktop hardware. The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. The cookie is used to store the user consent for the cookies in the category "Performance". This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". The cookie is used to store the user consent for the cookies in the category "Other. The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". The cookie is used to store the user consent for the cookies in the category "Analytics". These cookies ensure basic functionalities and security features of the website, anonymously. Necessary cookies are absolutely essential for the website to function properly. When you make a purchase using links on our site, we may earn an affiliate commission. Readers like you help support iGeeksBlog. 
WWDC 2022 wallpaper download: Make the wait worth it. 
|
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
